E01D0224 Human D Dimer ELISA kit
The Human D Dimer ELISA kit can be used to identify samples from the human species. D Dimer can also be called D2D, D 2 Dimer, D-Dimer.


E01D0224 Human D Dimer ELISA kit
The Human D Dimer ELISA kit can be used to identify samples from the human species. D Dimer can also be called D2D, D 2 Dimer, D-Dimer.
Product Information | |
Cat. No. | E01D0224 |
Product Name | Human D Dimer ELISA kit |
Species | Human |
Product Size | 48 Tests / 96 Tests |
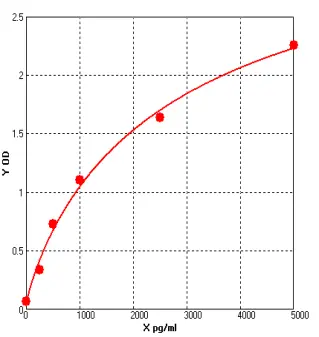
Concentration | 250-5000 pg/ml |
Sensitivity | 1.0 pg/ml |
Principal | Sandwich ELISA |
Sample Volume | 50 ul |
Sample Type | Serum, plasma, cell culture supernatants, body fluid and tissue homogenate |
Assay Time | 90 minutes |
Platform | Microplate Reader |
Conjugate | HRP |
Detection Method | Colorimetric |
Storage | 2-8°C |
Kit Components | ||
MATERIALS | SPECIFICATION | QUANTITY |
MICROTITER PLATE | 96 wells | stripwell |
ENZYME CONJUGATE | 10.0 mL | 1 vial |
STANDARD A (0.5mL) | 0 pg/mL | 1 vial |
STANDARD B (0.5mL) | 250 pg/mL | 1 vial |
STANDARD C (0.5mL) | 500 pg/mL | 1 vial |
STANDARD D (0.5mL) | 1000 pg/mL | 1 vial |
STANDARD E (0.5mL) | 2500 pg/mL | 1 vial |
STANDARD F (0.5mL) | 5000 pg/mL | 1 vial |
SUBSTRATE A | 6 mL | 1 vial |
SUBSTRATE B | 6 mL | 1 vial |
STOP SOLUTION | 6 mL | 1 vial |
WASH SOLUTION (100 x) | 10 mL | 1 vial |
BALANCE SOLUTION | 3 mL | 1 vial |
Principle of the Assay |
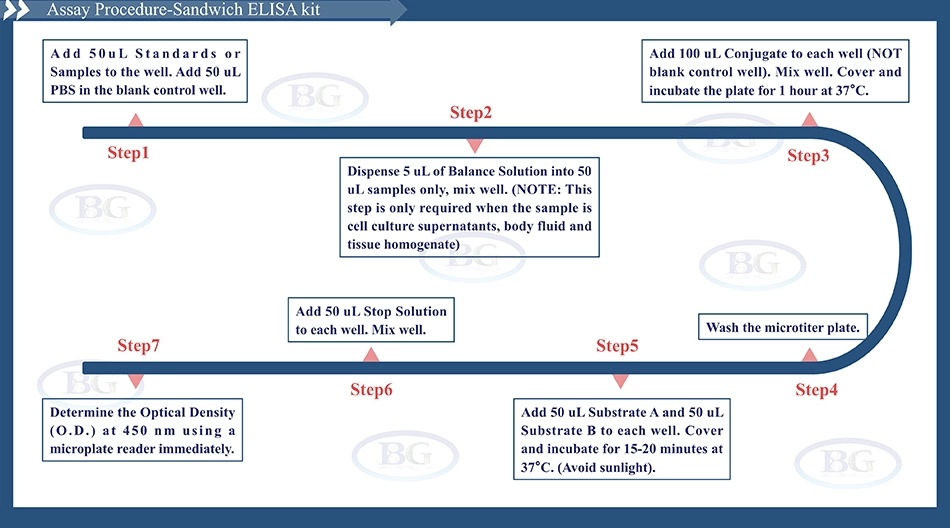
D2D ELISA kit applies the quantitative sandwich enzyme immunoassay technique. The microtiter plate has been pre-coated with a monoclonal antibody specific for D2D. Standards or samples are then added to the microtiter plate wells and D2D if present, will bind to the antibody pre-coated wells. In order to quantitatively determine the amount of D2D present in the sample, a standardized preparation of horseradish peroxidase (HRP)-conjugated polyclonal antibody, specific for D2D are added to each well to “sandwich” the D2D immobilized on the plate. The microtiter plate undergoes incubation, and then the wells are thoroughly washed to remove all unbound coD2Dnents. Next, substrate solutions are added to each well. The enzyme (HRP) and substrate are allowed to react over a short incubation period. Only those wells that contain D2D and enzyme-conjugated antibody will exhibit a change in color. The enzyme-substrate reaction is terminated by addition of a sulphuric acid solution and the color change is measured spectrophotometrically at a wavelength of 450 nm. A standard curve is plotted relating the intensity of the color (O.D.) to the concentration of standards. The D2D concentration in each sample is interpolated from this standard curve. |
Coefficient of Variance | Intra Variation% <10% | |
Inter Variation% <12% | ||
Recovery | 95-102% | |
Linearity | Diluent Ratio | Range % |
1:2 | 93-105 | |
1:4 | 88-106 | |
1:8 | 86-108 | |
Specificity/Cross-reactivity | No significant cross-reactivity or interference between D2D and analogues was observed. | |


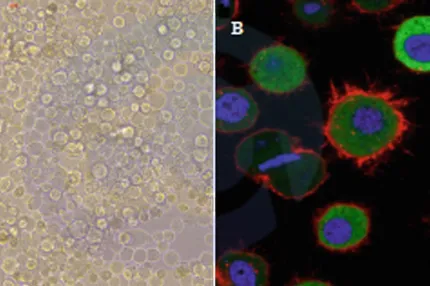
E01D0224 has been referenced in the below publications:
Study the role of vWF in assessing the Prethrombotic state and the severity of COPD in COPD patients.
The detection and significance of serum ICAM-1 and D-D in the patients with chronic severe hepatitis B and the studies of the influence on both with entecavir.
Type II Minimal-Invasive Extracorporeal Circuit for Aortic Valve Replacement: A Randomized Controlled Trial.
Related Bluegene Biotech Products
BlueGene Biotech News