BSA: Familiar Yet Unfamiliar – Launch of the Anti-Interference BSA ELISA Kit
Bovine Serum Albumin (BSA) is widely used in in vitro diagnostics and the industrial production of biopharmaceuticals, making it a familiar name in the field. However, its source, structure, function, and their correlations with applications remain relatively understudied. This article systematically elaborates on BSA in line with pharmacopoeial requirements to deepen understanding.
1. Composition of Plasma Proteins
Blood compositions are similar across different species. Whole blood with anticoagulants added, after centrifugation to remove blood cells, yields plasma as the supernatant. Plasma proteins are dynamic and complex mixtures of multiple proteins and molecular types, classified into Fractions I-V. Among these, Fraction II consists of globulins, Fraction IV of enzymes, and Fraction V of albumins – with Fractions II and V being the most abundant single-component fractions. Plasma proteins were first separated by electrophoresis by Tiselius in 1937, categorized into albumin, α, β, γ, and δ globulins.
With advancements in electrophoresis technology, Svensson and Longsworth[1,2] further subdivided plasma globulins into α1, α2, β1, β2, γ1, and γ2. In 1973, Laurell identified 13 major plasma proteins via agarose electrophoresis: albumin, lipoprotein, antitrypsin, transferrin, three complement proteins, fibrinogen, α1-acid glycoprotein, globin, IgG, IgA, and IgM[3,4,5]. Plasma proteins also include tissue-secreted enzymes such as acid/alkaline phosphatase, amylase, peptidases, and ribonucleases[6], as shown in Table 1:
Table 1
.png)
Human Serum Albumin (HSA) is a single-chain polypeptide folded into a heart-shaped structure. Its three-dimensional conformation comprises three domains, each containing two subdomains (A and B), with a total molecular weight of 66.8 kDa[7,8,9].
Albumin contains Sudlow's binding sites I/II, each formed by two loop structures, further constituting subdomains IIA and IIIA. Reported therapeutic compounds bind to the following two sites on HSA[10,11,12,13], as illustrated in Figures 1 and 2:
.png)
Figure 1
In essence, the amino acid compositions of albumin from human, bovine, and rat exhibit slight differences[14], as shown in Table 2. Mammalian albumins are characterized by low contents of methionine, glycine, and isoleucine, but high levels of cysteine, leucine, ionic amino acids, glutamic acid, and lysine. The abundance of ionic residues endows albumin with a high charge of 185 ions/molecule at pH=7, ensuring excellent solubility.
Albumin synthesized in the liver is not a glycoprotein; it consists solely of amino acids without prosthetic groups or other modifications. Even transthyretin (prealbumin, a precursor of albumin specialized in transporting retinol and thyroid hormone T4) in plasma contains almost no glycosyl groups and lacks the Asn-X-Ser/Thr motif required for N-glycosylation.
During circulation, albumin undergoes non-enzymatic glycosylation accumulation, with some "glycosylation modifications" being tightly bound substrates. Mixed cryptic disulfide bonds derived from cysteine and glutamine exert minimal effects on albumin unless oxidized and eliminated. In healthy humans, 1% of albumin forms covalent bonds with glucose. The glycosylation content of albumin has become a criterion for assessing its purity, with pure albumin requiring a glycosylation content of no more than 0.05% (w/w)[15]. The absence of sugars and tyrosine indicates acid hydrolysis of glycoproteins into black precipitates or humic substances. Albumin is typically colorless, but concentrated solutions bound to heme or bilirubin substrates exhibit a yellow hue.
Table 2
.png)
.png)
Figure 2
Metabolically, albumin is sufficiently large to avoid glomerular filtration and loss. Thus, it is utilized to transport fatty acids, steroids, bilirubin, porphyrins, and other therapeutic agents. It can also carry nanoparticles to deliver various macromolecular drugs to target cells/organs. For example, albumin-bound paclitaxel, approved by the FDA in 2005 for cancer treatment, uses albumin to deliver 130 nm particles and poorly water-soluble drugs to breast cancer tissues[16]. The active metabolite of non-steroidal anti-inflammatory drugs (NSAIDs), 6-methoxy-2-naphthoic acid, and 123I-labeled iodoamphetamine, circulate in plasma as albumin conjugates, cross the blood-brain barrier, reduce half-life, accumulate drugs in target tissues, and enable rapid clearance of radioactive elements[17,18], as shown in Figure 3:
.png)
Figure 3
Plasma transport proteins are diverse, each specialized in transporting specific substrates, as summarized in Table 3:
Table 3
.png)
Given the aforementioned functions, albumin is not only a key globulin for maintaining plasma osmotic pressure and nutritional balance but also a carrier for the transport of various plasma components. Therefore, human serum albumin is commonly used as a pharmaceutical agent for nutritional supplementation in surgical patients or as an excipient in drug formulations.
3. Diagnosis and Detection of BSA
BSA finds numerous applications in medical diagnostic sensors and enzymatic reactions. It serves as a protein reagent for measuring protein solutions (e.g., Bradford assay) and a standard protein in immunochemical assays due to its accuracy, rapidity, sensitivity, and reproducibility. Current BSA detection methods include electrochemical impedance spectroscopy, near-infrared reflectance spectroscopy, capillary electrophoresis, and light scattering.
Human plasma albumin and bovine plasma albumin share high structural similarity. BSA is a crucial marker for bovine health, milk quality, and meat production. It also has important applications in various industries, including pharmaceuticals, diagnostics, and food production. For instance, in cell culture, bovine serum – a key extract – maintains cell osmotic pressure and promotes cell division and growth. Additionally, BSA acts as a protective inert protein in various reagents to prevent target protein degradation and reduce interference from components in diagnostic samples.
Early capillary electrophoresis-based BSA detection leveraged the presence of tryptophan, tyrosine, and phenylalanine in BSA, enabling differential fluorescence-based detection[19]. Currently, protein quantification primarily relies on the Bradford method (total protein) and ELISA (specific BSA).
Recent pharmacopoeias clearly specify detection methods and concentration limits for added albumin. Notably, BSA contamination is common in laboratory settings due to its widespread use, requiring thorough equipment cleaning and dedicated experimental environments. We provide specialized manual washing solutions to ensure stable background signals.
· To meet pharmacopoeial requirements, Saitang Biotech has developed a Bovine Serum Albumin (BSA) ELISA Kit, with comprehensive performance validation including background signal, limit of detection (LOD), limit of quantitation (LOQ), precision, accuracy, and interference resistance. Key specifications:
· Calibration curve range: 0-100 ng/mL
· Background OD: 0.1-0.2
· S1/Blank ratio > 10
· LOD and LOQ: 0.0625 ng/mL and 0.5 ng/mL, respectively
· Precision (Intra & Inter CV): < 10% and < 15%, respectively
· Accuracy: 73.7%-108.3%
· Recovery rates in matrices such as 1% human serum, human albumin, and 1% gelatin: 70%-130%
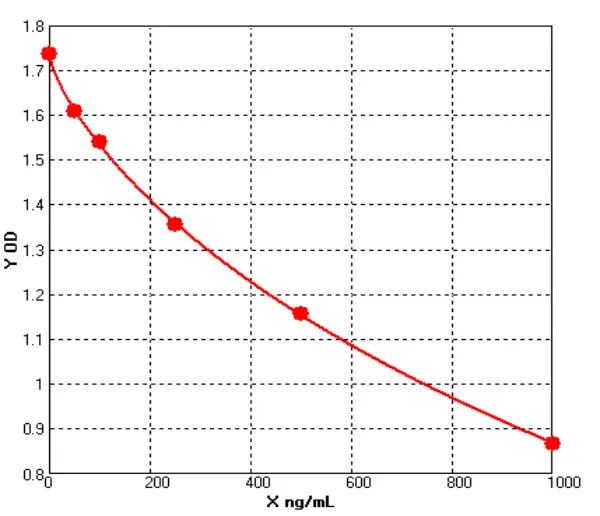
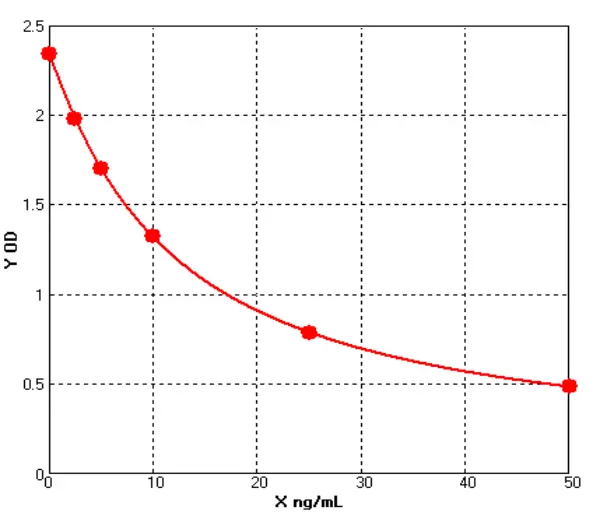
· See Figures 4-5 and Tables 4-5 for detailed data:
.png)
Figure 4
.png)
Figure 5
Table 4
.png)
Table 5
.png)
References:
[1] Svensson, H. Studies on Electrophoresis and Adsorption: I. The Moving Boundary Method in Electrophoresis and its Application to the Study of Proteins. Arkiv för Kemi, Mineralogi och Geologi. 1946. 22A (10):1-156.
[2] Longsworth, L. G. Diffusion Measurements, at 25℃, of Aqueous Solutions of Amino Acids, Peptides and Sugars. Chem. Rev. 1942. 30(2):323-340.
[3] Tiselius, A. A new apparatus for electrophoretic analysis of colloidal mixtures. Transactions of the Faraday Society. 1937. 33:524-531.
[4] Laurell C-B. Electroimmuno assay. Clin Chem. 1973. 19(1):99-102.
[5] R.L. Engle Jr., et al. The Plasma Proteins, 2, Academic Press, New York. 1960. pp184-265.
[6] Frank W Putnam. The Plasma Proteins:Structure, Function, and genetic control. New York. Second Edition / Volume I. 1975. p39.
[7] A. Jahanban-Esfahlan, et al. Spectroscopic and molecular docking studies on the interaction between N-acetyl cysteine and bovine serum albumin. Biopolymers. 2015. 103:638-645.
[8] J.K. Maurya, et al. A spectroscopic and molecular dynamic approach on the interaction between ionic liquid type gemini surfactant and human serum albumin, Journal of Biomolecular Structure and Dynamics. 2016. 34(10):2130-2145.
[9] N. Maurya, et al. In vitro cytotoxicity and interaction of noscapine with human serum albumin: Effect on structure and esterase activity of HSA. Molecular Pharmaceutics. 2019. 16(3) :952-966.
[10] A. Jahanban-Esfahlan, et al. Investigating the interaction of juglone (5-hydroxy-1, 4-naphthoquinone) with serum albumins using spectroscopic and in silico methods, Journal of the Iranian Chemical Society2017. 7(14):1527-1540.
[11] M. Kumari, et al. Effect of N-butyl-N-methyl-Morpholinium bromide ionic liquid on the conformation stability of human serum albumin. ChemistrySelect. 2017. 2(3):1241-1249.
[12] R. Patel, et al. Esterase activity and conformational changes of bovine serum albumin toward interaction with mephedrone: Spectroscopic and computational studies, Journal of Molecular Recognition. 2018. 31(11):e2734.
[13] Patel, M.U.H. et al. Spectroscopic and molecular modelling analysis of the interaction between ethane-1, 2-diyl bis (N, N-dimethyl-N-hexadecylammoniumacetoxy) dichloride and bovine serum albumin, Luminescence. 2015. 30(8):1233-1241.
[14] Theodore Peters Jr. The Albumin Molecule: Its Structure and Chemical Properties. Biochemistry, Genetics, and Medical Applications. 1995. p. 9-75, I-II.
[15] Hughes, W. L. et al. The Proteins. Academic Press, New York. 1954. 2:663.
[16] A. Jahanban-Esfahlan, et al. A simple improved desolvation method for the rapid preparation of albumin nanoparticles. International Journal of Biological Macromolecules. 2016. 91:703-709.
[17] Kawai, K., et al. Serum protein binding displacement: Theoretical analysis using a hypothetical radiopharmaceutical and experimental analysis with 123I-N-isopropyl-p-iodoamphetamine. Nuclear Medicine and Biology. 2009. 36(1):99e106.
[18] Rimac, H., et al. Displacement of drugs from human serum albumin: From molecular interactions to clinical significance. Current Medicinal Chemistry. 2017. 24(18):1930e1947.
[19] A. Jahanban-Esfahlan, et al. Interaction of glutathione with bovine serum albumin: Spectroscopy and molecular docking. Food Chemistry. 2016. 202:426-431
Cellgene Bioscience has been dedicated to the biopharmaceutical and industrial testing field for 15 years, offering a series of HCP residual detection products, as well as comprehensive technical services including HCP-specific antibody development and coverage analysis.
Cellgene Bioscience-Drug Residue Detection Products | |
Host Cell Protein ELISA kits (HCP) | |
CHO Host Cell Protein (CHO HCP) ELISA kit, G2 | |
CHO Host Cell Protein (CHO HCP) ELISA kit, G3 | |
HEK293 Host Cell Protein (HEK293 HCP) ELISA kit, G2 | |
E. coli Host Cell Protein (E. coli HCP) ELISA kit, G3 | |
Pichia pastoris Host Cell Protein (PP HCP) ELISA kit, G3 | |
Ogataea polymorpha Host Cell Protein ELISA kit, G3 | |
Saccharomyces cerevisiae Host Cell Protein ELISA kit, G3 | |
Spodoptera frugiperda (Sf9) Host Cell Protein ELISA kit, G3 | |
Medium Residues Detection kits | |
Protein A ELISA Kit (Boiling) | |
Protein A ELISA Kit | |
Mouse Immunoglobulin G ELISA Kit | |
Bovine Immunoglobulin G ELISA Kit | |
Human Immunoglobulin G ELISA Kit | |
Goat Immunoglobulin G ELISA Kit | |
Kanamycin ELISA Kit | |
Protein L ELISA Kit | |
NEGEP1271 | Protein G ELISA Kit |
Bovine Serum Albumin ELISA Kit | |
Human Serum Albumin ELISA Kit | |
Dextran Sulfate Salt Detection kit | |
Host Cell DNA Detection kits (HCD) | |
NS0 Host Cell DNA (NS0 HCD) Residue Detection kit | |
E.coli Host Cell DNA (E.coli HCD) Residue Detection kit | |
Vero Host Cell DNA (Vero HCD) Residue Detection kit | |
HEK293 Host Cell DNA (HEK293 HCD) Residue Detection kit | |
CHO Host Cell DNA (CHO HCD) Residue Detection Kit | |
Pichia Pastoris Host Cell DNA (PP HCD) Residue Detection Kit | |
Magnetic Residual DNA Sample Preparation Kit | |
Residual Total RNA Detection Kit | |
E.coli Residual Total RNA Detection Kit (qRT-PCR) | |
Host Cell Protein Antibodies | |
CHO Host Cell Protein G3 Antibody | |
CHO Host Cell Protein G2 Antibody | |
E.coli Host Cell Protein G3 Antibody | |
PH-E0021-2-Ab | Pichia Yeast Host Cell Protein G2 Antibody |
HEK293 Host Cell Protein G2 Antibody | |
Buffer Products | |
CG-H0100 | HCP ELISA buffer |
Protein L ELISA buffer | |